Ionic vs. Covalent
To determine if a compound is ionic or covalent you have to look on the periodic table.
Ionic Bonds
Ionic Bonds are formed between
a metal and nonmetal and they are
formed by transferring electrons. If
the compound has more than 2 elements,
it is Ionic.
Examples: CaO, HNO₃, NaCl, FrS, LiP
a metal and nonmetal and they are
formed by transferring electrons. If
the compound has more than 2 elements,
it is Ionic.
Examples: CaO, HNO₃, NaCl, FrS, LiP
Covalent Bonds
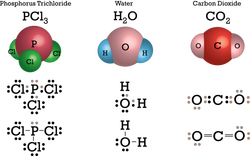
Covalent Bonds are formed between 2
non-metals and they are formed when
electrons are snared. The result compound
is called a molecular compound which
the molecule is the simplest form.
Examples: SO₂, P₂
non-metals and they are formed when
electrons are snared. The result compound
is called a molecular compound which
the molecule is the simplest form.
Examples: SO₂, P₂
